| |
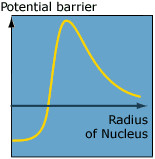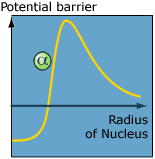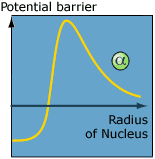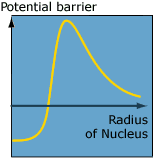 |
| In the classical world the positively
charged alpha particle needs enough energy to overcome
the positive potential barrier which originates from
protons in the nucleus. In the quantum world an alpha
particle with less energy can tunnel through the potential
barrier and escape the nucleus. |
|
|
|
Even though this required a
major rethink of our concept of reality, the Schrödinger
formulation of quantum mechanics became preferred by most
people, and about 10 years later, this probabilistic interpretation
of quantum mechanics became generally accepted. Schrödinger
also noted that his formulation was mathematically equivalent
to Heisenberg's – even though their interpretations
differed completely. In 1928, George Gamow published a theory
of radioactive 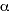 -decay based on a probabilistic quantum process,
where the -decay based on a probabilistic quantum process,
where the 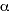 particle can 'tunnel' out of
the nucleus. particle can 'tunnel' out of
the nucleus. |