| |
 |
| Heisenberg dreamt up the gamma ray
microscope to explain his uncertainty principle. A source
of photons is used to illuminate an electron fired from
the left of the picture. The position of the electron
can be determined from the scattering of the photons
into the telescope at the bottom right of the picture. |
|
|
|
Heisenberg proposed a hypothetical
gamma ray microscope to illustrate his uncertainty principle.
The scattering of gamma rays is used to probe the position
of an electron as it traverses the microscope. The wavelength
'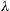 '
of the gamma rays is very small and so, they carry a large
amount of energy 'E', since E = h c / '
of the gamma rays is very small and so, they carry a large
amount of energy 'E', since E = h c / 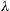 .
The trajectory of the electron will be perturbed by collisions
with the gamma ray probes. With Bohr’s help, Heisenberg
showed that the uncertainty in the measurement of the position
of the electron is approximately equal to the wavelength
of the gamma rays due to the effects of diffraction when
the gamma rays are viewed through the aperture of the telescope.
In other words .
The trajectory of the electron will be perturbed by collisions
with the gamma ray probes. With Bohr’s help, Heisenberg
showed that the uncertainty in the measurement of the position
of the electron is approximately equal to the wavelength
of the gamma rays due to the effects of diffraction when
the gamma rays are viewed through the aperture of the telescope.
In other words 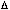 x ~ x ~ 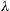 .
In the same fashion, the uncertainty on the momentum of
the electron is approximately equal to the momentum of the
single photon used to illuminate the electron, or, .
In the same fashion, the uncertainty on the momentum of
the electron is approximately equal to the momentum of the
single photon used to illuminate the electron, or, 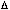 p ~h / p ~h / 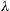 .
If these two uncertainties are multiplied together we obtain
the familiar uncertainty principle: .
If these two uncertainties are multiplied together we obtain
the familiar uncertainty principle:
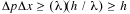
|