X-rays, what are they?

The penetrating rays discovered by Röntgen in 1895 - What Are They? Several important experiments around 1910 suggested that they are electromagnetic waves of shorter wavelength and higher energy than normal light. But the debates over the nature of the rays – waves or particles? – continued until the wave-particle duality was generally accepted in the 1920s. Photons can be described both as waves and particles.
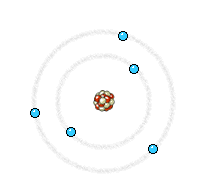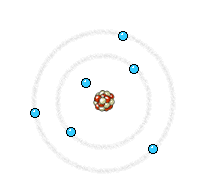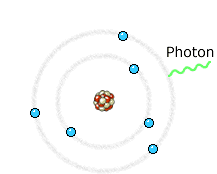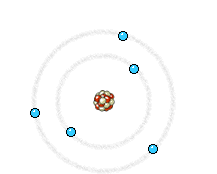
The electron (much lighter than the nucleus) comes very close to the nucleus and the electromagnetic interaction causes a deviation of the trajectory where the electron loses energy and an X-ray photon is emitted.
Bremsstrahlung X-rays
In an X-ray tube the electrons emitted from the cathode are accelerated towards the metal target anode by an accelerating voltage of typically 50 kV. The high energy electrons interact with the atoms in the metal target. Sometimes the electron comes very close to a nucleus in the target and is deviated by the electromagnetic interaction. In this process, which is called bremsstrahlung (braking radiation), the electron loses much energy and a photon (X-ray) is emitted. The energy of the emitted photon can take any value up to a maximum corresponding to the energy of the incident electron.
Characteristic X-ray lines
The high energy electron can also cause an electron close to the nucleus in a metal atom to be knocked out from its place. This vacancy is filled by an electron further out from the nucleus. The well defined difference in binding energy, characteristic of the material, is emitted as a monoenergetic photon. When detected this X-ray photon gives rise to a characteristic X-ray line in the energy spectrum. C. Barkla (The Nobel Prize in Physics 1917) observed these lines in 1908-09 and was given the 1917 Nobel Prize for this discovery. He also made the first experiments suggesting that the X-rays are electromagnetic waves.
X-rays behave like light!

Interference pattern observed by von Laue and collaborators using a photographic plate. The large central spot is due to the unscattered X-ray beam. The dark spots correspond to directions where X-rays scattered from different crystal (ZnS) layers interfere constructively.
Max von Laue (The Nobel Prize in Physics 1914) had the brilliant idea that if the X-rays are electromagnetic waves with a wavelength shorter than or of the same order as the separations between the ordered atoms in a crystal, then the scattering of X-rays against atoms could reveal some of the unknown properties of the X-rays. This is due to the interference effects that could be observed when an X-ray beam hits layers of atoms in a crystal. This effect is very similar to the interference of a ray of light hitting a gitter of densely packed scratches.
The experiment performed in 1912 with Röntgen's equipment was an immediate success and a direct proof that the X-rays are electromagnetic radiation. The wavelength observed was about 1000 times shorter than the wavelength of light. von Laue was given the 1914 Nobel Prize for his discovery of diffraction of X-rays.
X-rays Reveal crystal structures
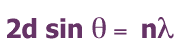
The Bragg formula
William Henry Bragg and his son William Lawrence (The Nobel Prize in Physics 1915) developed the method to analyze crystal structures using X-rays.
The famous Bragg formula
relates the wavelength ![]() of the X-ray with
the distance d between crystal planes and the
incident angle of the X-ray
of the X-ray with
the distance d between crystal planes and the
incident angle of the X-ray ![]() giving rise to constructive interference.
giving rise to constructive interference.
For their analysis of crystal structures they were awarded the 1915 Nobel Prize.
More about the Bragg formula
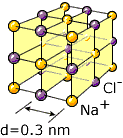
Crystal planes, in NaCl, ordinary salt. Other planes are also possible.
X-rays scattered from different layers of atoms can interfere with each other. The interference depends on the wavelength of the X-ray and on the distance between the atom layers. An X-ray with well-known wavelength can be used to explore the structure of the crystal. For a well-known crystal, the X-ray properties can be examined.
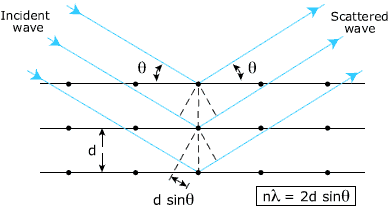
X-ray scattering from three
crystal planes, separated by the distance d. For
constructive interference in a direction ![]() the path difference must be an even number of
wavelengths.
the path difference must be an even number of
wavelengths.
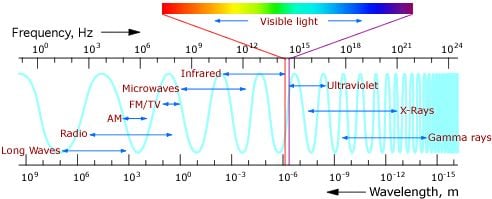
The electromagnetic spectrum
X-rays have wavelengths much shorter than visible light, but longer than high energy gamma rays. Their wavelength is well suited to study crystal structures and details of the human body. In addition, several objects and processes in the Universe emit X-rays. These X-rays are the messangers revealing information of the cosmos.
MLA style: "X-rays". Nobelprize.org. Nobel Prize Outreach AB 2022. <http://nobel-external-educationalgames-app.azurewebsites.net/educational/physics/x-rays/what.php>