History and Properties of Liquid Crystals
An Entirely New Phase of Matter
 |
A liquid crystal |
In 1888, the Austrian chemist Friedrich Reinitzer, working in the Institute of Plant Physiology at the University of Prague, discovered a strange phenomenon. Reinitzer was conducting experiments on a cholesterol based substance trying to figure out the correct formula and molecular weight of cholesterol. When he tried to precisely determine the melting point, which is an important indicator of the purity of a substance, he was struck by the fact that this substance seemed to have two melting points. At 145.5°C the solid crystal melted into a cloudy liquid which existed until 178.5°C where the cloudiness suddenly disappeared, giving way to a clear transparent liquid. At first Reinitzer thought that this might be a sign of impurities in the material, but further purification did not bring any changes to this behavior.
Puzzled by his discovery, Reinitzer turned for help to the German physicist Otto Lehmann, who was an expert in crystal optics. Lehmann became convinced that the cloudy liquid had a unique kind of order. In contrast, the transparent liquid at higher temperature had the characteristic disordered state of all common liquids. Eventually he realized that the cloudy liquid was a new state of matter and coined the name "liquid crystal," illustrating that it was something between a liquid and a solid, sharing important properties of both. In a normal liquid the properties are isotropic, i.e. the same in all directions. In a liquid crystal they are not; they strongly depend on direction even if the substance itself is fluid.
This new idea was challenged by the scientific community, and some scientists claimed that the newly-discovered state probably was just a mixture of solid and liquid components. But between 1910 and 1930 conclusive experiments and early theories supported the liquid crystal concept at the same time that new types of liquid crystalline states of order were discovered.
At the early time of Reinitzer and Lehmann, scientists only knew three states of matter. The general idea was that all matter normally had one melting point, where it turns from solid to liquid, and a boiling point where it turns from liquid to gas. Water is a good example of this view. It melts at 0°C and it boils and becomes steam, which is water in its gaseous form, at 100°C.
Today, thanks to Reinitzer, Lehmann and their followers, we know that literally thousands of substances have a diversity of other states. Some of them have been found very usable in several technical innovations, among which liquid crystal screens and liquid crystal thermometers may be the best known.
 |
| Pierre-Gilles de Gennes |
In the 1960s, a French theoretical physicist, Pierre-Gilles de Gennes, who had been working with magnetism and superconductivity, turned his interest to liquid crystals and soon found fascinating analogies between liquid crystals and superconductors as well as magnetic materials. His work was rewarded with the Nobel Prize in Physics 1991. The modern development of liquid crystal science has since been deeply influenced by the work of Pierre-Gilles de Gennes.
What is So Special about Liquid Crystals?
Liquid crystals are partly ordered materials, somewhere between their solid and liquid phases. Their molecules are often shaped like rods or plates or some other forms that encourage them to align collectively along a certain direction. The order of liquid crystals can be manipulated with mechanical, magnetic or electric forces. Finally, liquid crystals are temperature sensitive since they turn into solid if it is too cold, and into liquid if it is too hot. This phenomenon can, for instance, be observed on laptop screens when it is very hot or very cold.
 |
Liquid Crystal Phases
 |
 |
| The twist in a cholesteric liquid crystal | Liquid crystal in the smectic C phase |
Liquid crystals have two main
phases which are called the "nematic phase" and the
"smectic phase."
The nematic phase is the simplest of liquid crystal
phases and is close to the liquid phase. The
molecules float around as in a liquid phase, but are
still ordered in their orientation. When the
molecules are chiral* and in the nematic phase, they
arrange themselves into a strongly twisted structure
that often reflects visible light in different bright
colors which depend on the temperature. They can
therefore be used in temperature sensors
(thermometers). This special case of a nematic is
often called cholesteric. The name is historic as it
goes back to the substances on which Reinitzer made
his discovery.
The smectic phase is close to the solid phase. The
liquid crystals are here ordered in layers. Inside
these layers, the liquid crystals normally float
around freely, but they cannot move freely between
the layers. Still, the molecules tend to arrange
themselves in the same direction. The smectic phase
is in turn divided into several sub-phases with
slightly different properties.
* The definition of chirality is "something that can not be superimposed on its own mirror image, like a hand."
How Do Liquid Crystal Displays Work?
To understand how a liquid crystal screen works, you must first understand the concept of light polarization. Light is made out of particles called photons. These photons travel at the speed of light. While moving, a photon vibrates in a plane which is perpendicular to its direction, but within this plane the vibration direction is random for normal (non-polarized) light.
 |
 |
| These four arrows represent unpolarized light rays. As you can see, the arrows are tilted in random directions | Four parallel arrows representing polarized light |
Some processes affect the
direction of vibration. For instance, if sunlight is
reflected off the surface of a road, or the surface
of the open sea, the reflected light contains more
vibrations parallel to this surface than
perpendicular to it. The light has been polarized, in
this case only partly. Some materials however, like
the plastic used in polarizing sunglasses, may absorb
almost all vibration components along a certain
direction and only let through vibrations along the
perpendicular direction. In normal sunglasses the
admitted vibration direction is vertical. This is why
polarizing sunglasses can be used to remove the glare
of reflected light from the surface of a road, or the
surface of the open sea.
To polarize light you can use such a polarization
filter as found in sunglasses. Two such polarization
filters, placed after each other along the light with
their admitting direction perpendicular to each other
will not let any light through.
So what has this to do with liquid crystal displays? Well, the conventional liquid crystal display basically consists of such a package of two crossed polarizers with a liquid crystal in between. If the molecules lie perpendicular to the plane of the polarizers, i.e. along the direction of the light ray, they have no influence on the state of polarization. Thus, the package of crossed polarizers lets no light through. The cell appears black. On the other hand, if the molecules are arranged to lie parallel to the plane of the polarizers, i.e. in the plane in which the light vibrates, the presence of the liquid crystal will strongly affect the state of polarization.
In the so-called twisted nematic (or TN) display, the molecules are arranged in this way. More specifically, the glass surfaces are treated such that the molecular direction is parallel to the admitting direction of each neighboring polarizer. Because these directions are crossed, the molecular direction is confined to a 90° twist from one side of the cell to the other (see figure).
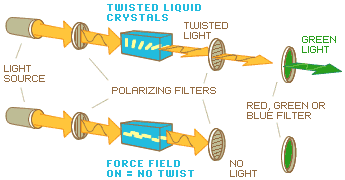
In this case, what happens is
that the light vibration follows this twist from one
polarizer to the other, so that all light in fact
passes the cell, without being absorbed, in spite of
the fact that the polarizers are crossed. Hence, the
cell appears bright.
As mentioned earlier, liquid crystals are sensitive
to electric forces. If you apply an electric field
that is strong enough across a liquid crystal of the
right kind, its molecules arrange themselves parallel
to the electric field. So now, by applying a voltage
across the liquid crystal cell, i.e. along the light
direction, you destroy the twist and instead force
the molecules into the direction in which they do not
affect the polarization state of the light. All light
is now absorbed by the crossed polarizers and the
cell appears black when the electric field is turned
on.
By creating a matrix of squares that locally control the state of the twist in their respective area you get a liquid crystal display containing a large number of individual picture elements (pixels).

First published 9 September 2003
| Related Laureate | Tips | |
in Physics 1991 - Pierre-Gilles de Gennes » |
Play the liquid crystal games » | |